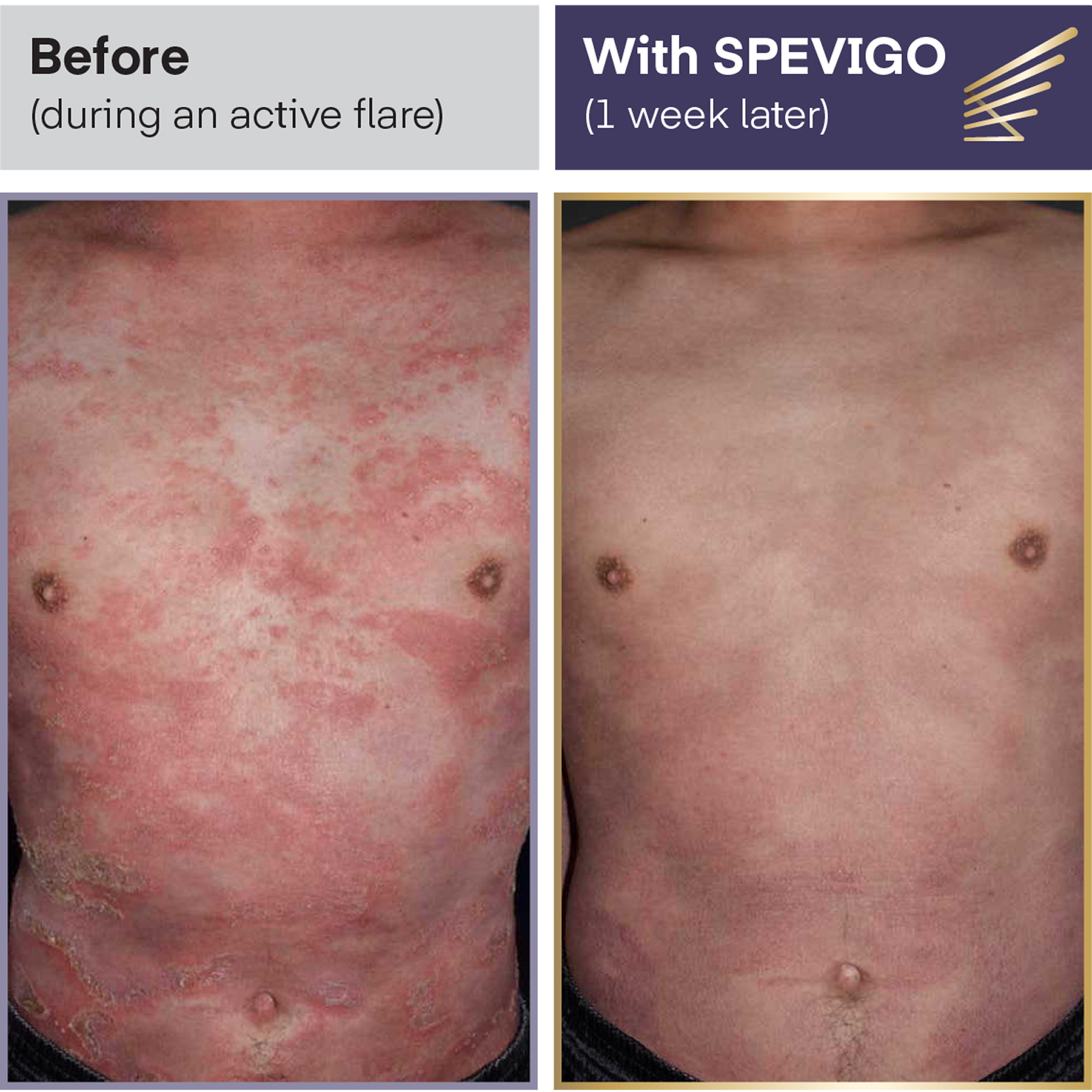
Clear results:
complete pustular clearance within 1 WEEK

54% of people treated with SPEVIGO vs 6% of people taking placebo
19 out of 35 people on SPEVIGO vs 1 out of 18 on placebo

This photo represents an individual from the SPEVIGO IV clinical trial, which measured how many people had no visible pustules 1 week after treatment, as well as safety, in people experiencing an active GPP flare.
Pustular clearance as early as DAY 2*
11% of people treated with SPEVIGO (4 out of 35) had no visible pustules vs no one (0 out of 18) taking placebo
These results are exploratory and were not the main purpose of the study, meaning they indicate a possible effect of SPEVIGO, and the results are less certain.
Individual results may vary based on many factors, including elements like the severity of your GPP, your treatment plan, and any other conditions you may have.
*These results are based on a clinical trial of 53 people with GPP.
“Placebo” means no active medication.

Support for your next steps
This guide can help you prepare for a conversation with your doctor, including common questions to consider.
Hear from a doctor on the impact of SPEVIGO
Listen to Dr Chovatiya explain the results when people received SPEVIGO in clinical trials.

Dr Chovatiya has been compensated for his participation.
GPP=generalized pustular psoriasis.
IV=intravenous.
SC=subcutaneous.